In applying the modeling methodology to IBDP physics, there are a few gaps. In this post, I present a unit that uses the modeling approach for the thermal physics and thermodynamics unit of Physics.
1. Building background
Have students touch something warm and sonething cold. Ask what is happening in terms of energy in these interactions. There are two main goals for this short preliminary discussion:
a. definitions of the terms heat and internal energy
b. an agreement that heat, as a transfer of energy, is sufficient to explain thermal processes, and is what we feel
2. Paradigm Lab I
For this lab, masses of hot and cold water are mixed in an insulated cup. After a demonstration of the effect, students should walk through a variable brainstorming session (I like a version I learned from Karl Schmidt that has three steps: observations, measure-ables, manipulate-ables). The students should recognize that either the hot water mass or the cold water mass could serve as the independent variable. They will recognize that the temperatures are dependent, but might need some help to decide to use the ratio of temperature changes as the dependent variable.
The students will thus conduct two labs, perhaps by splitting into two groups and sharing in the whiteboard meeting. They should find that the ratio of temperature changes is proportional to one of the masses and inversely proportional to the other. Be careful to define the ratio of temperature changes clearly for everyone before beginning data collection.
Combining results and looking at slopes allows us to construct the equation:
mass of hot water * temperature change for hot water = mass of cokd water * temperature change for cold water
Now, the students should recognize that this reflects the heat flow discussed earlier, but since heat is n energy change the units don’t work out and we need to have a coefficient. Students should think about what this coefficient means (it is the energy released/absorbed to change the temperature by one degree Celcius for each kilogram of matter).
3. Some practice with calorimetry problems without phase changes here. I spontaneously put two on the board at the front, and had student pairs write a third.
4. Demonstrate a melting curve. This may be difficult, but at least set up the apparatus and sketch the resulting curve.
First provide students with the power output of the heater and the mass of your sample and ask them to find the specific heat capacity of the sample (ie: using the slope).
Next, point to a segment where the temperature is unchanging and ask students to try to explain what is happening here. Socratic questioning and your favourite applet (PhET, etc) is good here. There are lots of misconceptions here, so insist on very clear statements and model IB language (ie: from past exams).
Students can usually guess the form of the latent heat equation (Q=mL), so I ask them how they would design an experiment to test that, and then we move on.
5. Model deployment, proper.
Now, the students are ready for some lengthier calorimetry problems, so they do a worksheet of these.
6. Quiz
A quiz based on the worksheet.
7. Practical I
I do a calorimetry problem at the front of the room, moving a hidden mass of steel from a hot water bath to a known mass of cold water. I provide the temperatures and the students predict the mass, which we put on a balance once all the predictions are in.
8. Paradigm Lab II
This could be done using a PhET simulation, or with an apparatus like the one shown below (the block could be replaced with a heating element to manipulate the temperature, and a pressure sensor should be attached to the flask somewhere):
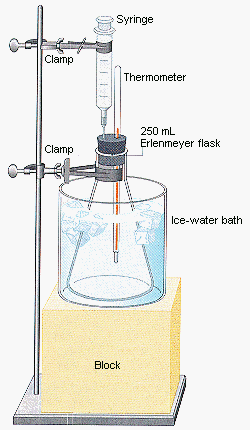
Image from Purdue University (http://chemed.chem.purdue.edu/)
Briefly outline the nature of an ideal gas (no potential energy, elastic collisions providing pressure, etc). In the regular pre-lab process, students should identify pressure, volume, temperature, and number of molecules (or mols) as the relevant variables. For now, leave the number of moles aside (ie: keep the device closed): this leaves three experiments, all of which should be done:
- How does a changing temperature affect the pressure at constant volume?
- How does a changing temperature affect the volume, when the volume is adjusted (ie: by extending the syringe) so that the pressure is constant?
- How a changing volume affect the pressure at constant temperature? (this works better by connecting the syringe directly to the pressure sensor)
Connecting these gives that PV/T = constant. Logically, P and V should be proportional to the number of moles n, while T is inversely proportional to n (at constant P and V), so this becomes PV/T = nR, where R is the ideal gas constant.
After rearranging this equation into the familiar form (PV=nRT), as students to determine the units on the left side. This gives the units of R as J/mol/K and, more importantly, shows that the ideal gas law is fundamentally a statement about the amount of internal energy in the gas. A brief note about the Maxwell distribution and rms velocities of the particles is probably appropriate here, even though it does take us out of the modeling cycle.
9. Deployment II
Now, the students do a second worksheet, on the ideal gas law. This also includes the three constituent laws (Boyle’s, Charles’, and the Gay-Lussac/Amonton) and practice with the exam-style question “determine whether this graph shows that the gas obeys Boyle’s Law (etc)” by checking points on the graph to determine whether the relationship is proportional, inverse, or otherwise.
10. Practical II
11. Unit Test